On
The lawsuits, deposited in the
"We are filing these lawsuits to protect the innovative mRNA technology platform that we pioneered, invested billions of dollars in creating, and patented during the decade preceding the COVID-19 pandemic," stated
Though Intellectual Property (IP) disputes are not uncommon in the pharmaceutical industry, the scale and prominence of this litigation are of rare magnitude.
What is mRNA technology?
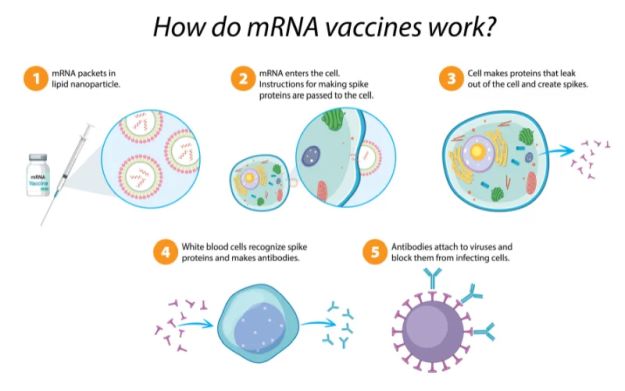
At the heart of the IP dispute are immunological techniques that utilize mRNA - short for "messenger ribonucleic acid." Unlike DNA, which has a familiar double-helical arrangement, RNA encodes genetic information in a single, looped strand and is the primary structure in virus genomes. As its name implies, mRNA is used in more complex cells to relay genetic information, specifically, during protein synthesis. In essence, the mRNA molecule acts as a short-lived "burn after reading" data packet that is first transcribed from DNA, transferred within the cell, read, translated to produce the corresponding protein and then degraded.
Synthetic-mRNA vaccines exploit this mechanism to cause the patient's body to produce viral spike proteins. Despite being neither a virus nor dangerous, these identifying spike proteins - called antigens - trigger the body's normal immune response and are destroyed. Subsequently, the immune system retains a memory of the encountered antigen and is able to recognize and respond to it more quickly in the event of an actual viral infection.
Not all vaccines for COVID-19 rely on mRNA processes. For instance, the Oxford-AstraZeneca COVID-19 vaccine uses the older and more pervasive method of delivering antigens by way of a modified virus. In this approach, a weakened or "dead" virus is injected into the patient in order to stimulate an immune response - the antigens being present in the dose. In an mRNA vaccination, the body is fooled into producing the required antigens itself by artificial "blueprints." Both methods have been proven to be highly effective.
Prior to the expedited approval of the first COVID-19 vaccines by national regulators in late 2020, no pharmaceutical product based on mRNA technology had seen public deployment. Curiously, the Pfizer-BioNTech and
Why now?
In
As such, the unspecified damages sought from
In its own statement,
Both
Patents in medicine
The purpose of the patent regimes in pharmaceutical research is to secure sufficient compensation to recoup extremely high development costs and fund additional projects. As such,
Dennemeyer's legal experts in
The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances.
Dennemeyer
55, rue des Bruyeres
1274 Howald
Luxembourg
LUXEMBOURG
Tel: 368403609
Fax: 368403610
E-mail: crupp@dennemeyer.com
URL: www.dennemeyer.com
© Mondaq Ltd, 2022 - Tel. +44 (0)20 8544 8300 - http://www.mondaq.com, source








