(via TheNewswire)
 | |||||||||
OVERVIEW
The Company reported a netloss and total comprehensive lossof
Clinical development
LDOS47 in lung cancer
The Phase I study combination therapy in lung cancer (LDOS001) was completed and the final clinical trial report issued in
December 2021 . A manuscript was accepted for publication inJournal of Thoracic Oncology Clinical and Research Reports onSeptember 2 nd, 2022.The Phase II study of combination therapy in lung cancer (LDOS003) was halted in the dose escalation portion of the study in 2020 at the height of pandemic lockdown and the final abbreviated clinical trial report has been further delayed amidst war in
Ukraine where all subjects were recruited. A final effort is being made in an attempt to retrieve any data that is salvageable and conclude the study.Another important aspect of the development of the LDOS47 platform is the combination with chemo- and/or immuno-therapy, that may boost the utility of the platform. The Company has engaged several key opinion leaders to evaluate the feasibility of such combinations and aims to develop a roadmap by the second quarter of 2023.
LDOS47 in pancreatic cancer
The Company's Phase Ib/II combination trial in pancreatic cancer (LDSOS006) continues to recruit patients, as the Company remains committed to this study. The second dosing cohort was successfully completed in
Corporate development
On
August 9, 2022 , the Company announced that it has entered into a two-year scientific collaboration agreement (“Agreement”) with University Hospital Tubingen (Germany ) to assess the therapeutic response of L-DOS47 in several cancer models expressing CEACAM6, with advanced preclinical metabolic imaging.
On
August 30, 2022 , the Company announced that it had completed the buyback of the outstanding amount of the convertible security funding agreement withLind Global Macro Fund, LP . The Company entered into the Agreement with Lind inMay 2021 and closed the first tranche under the Agreement for gross proceeds of$3,500,000 shortly thereafter. The Company has now bought back the amount outstanding of the Convertible Security under the Agreement, which isC$2,061,875 .
On
September 1, 2022 , the Company announced the appointment of Dr. Gabrielle M Siegers, MA, Ph.D., as the Head of RD based out of the Company’s lab inEdmonton .
On
September 12, 2022 , the Company applied to the TSX to price protect a proposed$5 million financing of common shares at a price of$0.18 per share. The TSX granted a price protection letter onSeptember 14, 2022 , and the conditional approval of the placement onSeptember 26, 2022 . As ofOctober 31, 2022 , the Company has received a total of$4,629,020 in subscription receipts related to this financing with insiders subscribing for$270,000 . OnNovember 3, 2022 , the Company announced that it had closed a private placement financing for net proceeds of CAD$4,629,020 from the issuance of 25,716,777 common shares at a price of$0.18 per common share. The common shares issued pursuant to the Private Placement are subject to a statutory hold period of four months and one day ending onMarch 4, 2023 , in accordance with applicable securities law. In connection with the closing, the Company paid a cash fee of 10% of gross proceeds raised to an eligible finder.
On
October 3, 2022 , the Company announced the appointment of Dr.Frank Gary Renshaw , as the Chief Medical Officer.
On
December 8, 2022 , the Company announced the appointment of Mr.Jacek Antas as the CEO of Helix replacing Mr.Artur Gabor who had resigned as the CEO and as a director of the Company. Furthermore, Ms.Malgorzata Laube was appointed to the Board of Directors of the Company with immediate effect.
Research and development
Research & development expenses for the three-month period ended
The following table outlines research and development costs expensed for the following periods:

Click Image To View Full Size
Research and development expenditures for the three-month period ended
Operating, general and administration
Operating, general and administration expensesfor the three-month period ended
The following table outlines operating, general and administration expenses for the following periods:

Click Image To View Full Size
Operating, general and administrationexpenditures for the three-month period ended
Since
Wages and benefits were decreased by
LIQUIDITY AND CAPITAL RESOURCES
The Company reported a net loss and total comprehensive loss for thethree-month periods ended
On
During the three-month period ended
In order for the Company to advance the currently planned preclinical and clinical research and development activities, its collaborative scientific research programs and pay for its overhead costs, the Company will need to raise approximately
The Company currently has three clinical studies (seeResearch and Development Activitiesabove for additional information) in various stages. The Company has completed the clinical study report for LDOS001 and submitted a final annual report to the FDA in
The Company received IND approval by the FDA to conduct a Phase I-b/II study (LDOS006) in the
The Company is also forecasting
The Company’sStatement of Financial Position andStatement of Net Loss and Comprehensive Loss for the three-month periods ended
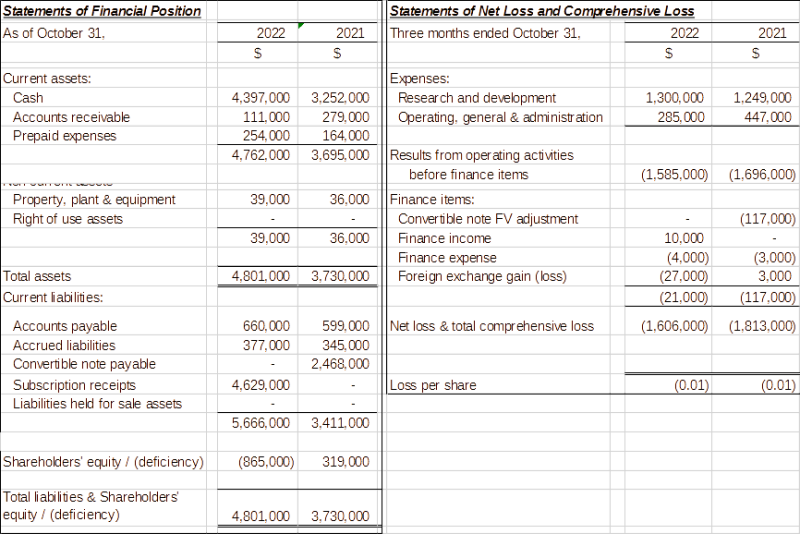
Click Image To View Full Size
The Company’s financial statements, management’s discussion and analysis and annual information form will be filed under the Company’s profile on SEDAR atwww.sedar.com, as well as on the Company’s website atwww.helixbiopharma.com.
About
Company Contact
Tel: 905-841-2300
namrata@grovecorp.ca
Forward-Looking Statements and Risks and Uncertainties
This news release contains forward-looking statements and information (collectively, “forward-looking statements”) within the meaning of applicable Canadian securities laws. Forward-looking statements are statements and information that are not historical facts but instead include financial projections and estimates, statements regarding plans, goals, objectives, intentions and expectations with respect to the Company’s future business, operations, research and development, including the focus of the Company’s primary drug product candidate L-DOS47 and other information relating to future periods.
Forward-looking statements include, without limitation, statements concerning (i) the Company’s ability to operate on a going concern being dependent mainly on obtaining additional financing; (ii) the Company’s priority continuing to be L-DOS47; (ii) the Company’s development programs, clinical studies, trials and reports for DOS-47 and L-DOS47; (iii) the Company’s development programs for DOS47 and L-DOS47; (iv) future expenditures, the insufficiency of the Company’s current cash resources and the need for financing; (v) future financing requirements,and the seeking of additional funding, and (vi) forecasts and future projections regarding development programs and expenditures. Forward-looking statements can further be identified by the use of forward-looking terminology such as “ongoing”, “estimates”, “expects”, or the negative thereof or any other variations thereon or comparable terminology referring to future events or results, or that events or conditions “will”, “may”, “could”, or “should” occur or be achieved, or comparable terminology referring to future events or results.
Forward-looking statements are statements about the future and are inherently uncertain and are necessarily based upon a number of estimates and assumptions that are also uncertain. Although the Company believes that the expectations reflected in such forward-looking statements are reasonable, such statements involve risks and uncertainties, and undue reliance should not be placed on such statements. Forward-looking statements, including financial outlooks, are intended to provide information about management’s current plans and expectations regarding future operations, including without limitation, future financing requirements, and may not be appropriate for other purposes. Certain material factors, estimates or assumptions have been applied in making forward-looking statements in this news release, including, but not limited to, the safety and efficacy of L-DOS47; that sufficient financing will be obtained in a timely manner to allow the Company to continue operations and implement its clinical trials in the manner and on the timelines anticipated; the timely provision of services and supplies or other performance of contracts by third parties; future costs; the absence of any material changes in business strategy or plans; and the timely receipt of required regulatory approvals and strategic partner support.
The Company’s actual results could differ materially from those anticipated in the forward-looking statements contained in this news release as a result of numerous known and unknown risks and uncertainties, including without limitation, the risk that the Company’s assumptions may prove to be incorrect; the risk that additional financing may not be obtainable in a timely manner, or at all, and that clinical trials may not commence or complete within anticipated timelines or the anticipated budget or may fail; third party suppliers of necessary services or of drug product and other materials may fail to perform or be unwilling or unable to supply the Company, which could cause delay or cancellation of the Company’s research and development activities; necessary regulatory approvals may not be granted or may be withdrawn; the Company may not be able to secure necessary strategic partner support; general economic conditions, intellectual property and insurance risks; changes in business strategy or plans; and other risks and uncertainties referred to elsewhere in this news release, any of which could cause actual results to vary materially from current results or the Company’s anticipated future results. Certain of these risks and uncertainties, and others affecting the Company, are more fully described in the Company’s interim management’s discussion and analysis for the three-month period ended
__________
Copyright (c) 2022 TheNewswire - All rights reserved.
Copyright (c) 2022 TheNewswire - All rights reserved., source






